|
Water, formally known as dihydrogen monoxide, can be produced by combining two molecules of diatomic hydrogen (H2) and one molecule of diatomic oxygen (O2). This process results not only in the production of two water (H2O) molecules, but also in the release of energy. Known for over two centuries, this reaction describes what happens in a hydrogen fuel cell where hydrogen and oxygen are used as fuel to create water and energy.
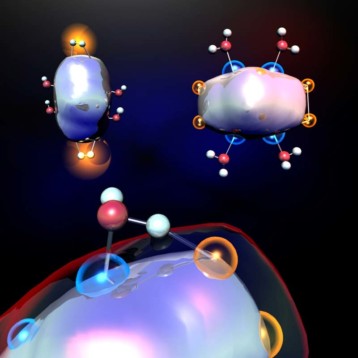
In a typical hydrogen fuel cell, the diatomic hydrogen gas enters one side of the cell as the diatomic oxygen gas enters the other side. The hydrogen molecules undergo oxidation, meaning they lose their electrons to become positively charged, while the oxygen molecules undergo reduction, or in other words, gain the extra electrons to become negatively charged. The negatively charged oxygen ions combine with the positively charged hydrogen ions to form water, releasing electrical energy in the process.
The UI scientists managed to simplify the oxygen reduction, considered to be the more complicated side of the reaction, through the use of unconventional metal hydrides. Thomas Rauchfuss, Professor of Chemistry at the University of Illinois, and Zachariah Heiden, a doctoral student at the University, investigated unconventional metal hydrides for oxygen reduction. In their paper, recently published in the Journal of American Chemical Society, they focused exclusively on the oxidative reactivity of iridium based transfer and hydrogenation catalysts in a homogenous, non aqueous solvent.
While most catalysts react with either hydrogen or oxygen, iridium reacts with both in a homogenous, non aqueous environment to form a hydride. The hydride then reacts with oxygen to produce water. This new catalyst could lead to the creation of more efficient hydrogen fuel cells, substantially lowering their cost.
You can read more about iridium as a catalyst here.