|
The traditional methods used to treat patients with cancer are problematic because they usually involve large dosage of drugs that have life-threatening side effects. Now, a team of engineers from the Massachusetts Institute of Technology (MIT) devised a way to deliver the necessary drugs by “smuggling” them on the backs of the cells sent in to fight the tumor. That way, the drugs reach only their intended targets, greatly reducing the risk to the patient.
“What we’re looking for is the extra nudge that could take immune-cell therapy from working in a subset of people to working in nearly all patients, and to take us closer to cures of disease rather than slowing progression,” says Darrell Irvine, the senior author of the study.
The paper, recently published in Nature Medicine, presents a new approach that could dramatically improve the success rate of immune-cell therapies, which hold promise for treating many types of cancer. Furthermore, the new method could also be used to deliver other types of cancer drugs or to promote blood-cell maturation in bone-marrow transplant recipients, according to the researchers.
The research details immune-cell therapy, which requires removing a type of immune cells called T cells from the patient, engineering them to target the tumor, and injecting them back into the patient. Those T cells then hunt down and destroy tumor cells. Currently clinical trials are conducted for ovarian and prostate cancers, as well as melanoma.
“The major limitation right now is getting enough of the T cells that are specific to the cancer cell,” said Glenn Dranoff, associate professor of medicine at Harvard Medical School. So, although immune-cell therapy is a very promising approach to treating cancer, getting it to work has proved challenging. According to Dranoff, the problem is getting T cells to function properly in the patient.
Many researchers have tried to overcome those obstacles; one of the efforts described in previous studies is injecting patients with adjuvant drugs that stimulate T-cell growth and proliferation. One class of drugs tested in clinical trials is interleukins — naturally occurring chemicals that help promote T-cell growth but have severe side effects, including heart and lung failure, when given in large doses.
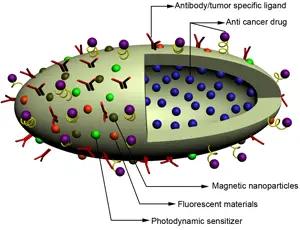
In this new study, Irvine and his colleagues chose a different path. They decided to avoid toxic side effects by designing drug-carrying pouches made of fatty membranes that can be attached to sulfur-containing molecules normally found on the T-cell surface. The researchers injected T cells, each carrying about 100 pouches loaded with the interleukins IL-15 and IL-21, into mice with lung and bone marrow tumors. Once the cells reached the tumors, the pouches gradually degraded and released the drug over a weeklong period. The drug molecules attached themselves to receptors on the surface of the same cells that carried them, stimulating them to grow and divide.
The results were encouragingly positive: within 16 days, all of the tumors in the mice treated with T cells carrying the drugs disappeared. Those mice survived until the end of the 100-day experiment, while mice that received no treatment died within 25 days, and mice that received either T cells alone or T cells with injections of interleukins died within 75 days.
Dranoff – which was not part of the study – compliments this innovative approach. “The idea of modifying T cells in the lab to make them work better is something many people are exploring through more complicated approaches such as gene modification,” he said. “But here, the possibility of just attaching a carefully engineered nanoparticle to the surface of cells could be a much simpler procedure.”
Although the inquiry is far from over, Irvine already thinks about future benefits derived from this technique, such as targeted delivery of chemotherapy agents. “There are lots of people studying nanoparticles for drug delivery, especially in cancer therapy, but the vast majority of nanoparticles injected intravenously go into the liver or the spleen,” he said. “Less than 5 percent reach the tumor.”
The new way to carry drugs specifically to tumors could provide scientists a way to resurrect promising drugs that failed in clinical trials because they were cleared from the bloodstream before they could reach their intended targets, or had to be given in doses so high they had toxic side effects. Furthermore, delivering drugs that accelerate blood-cell production along with the bone-marrow transplant could shorten the period of immunosuppression, making the process safer for patients, said Irvine.
The next step for Irvine and his team is making sure the manufacturing process will yield nanoparticles safe to test in humans. Once that is done, they hope the particles could be used in clinical trials in cancer patients, possibly within the next two or three years.
TFOT has also covered another “smart” nanocapsule delivery system, developed at the UCLA Henry Samueli School of Engineering and Applied Science, and the development of a new contrast agent to help fight Cancer, made at Delft University of Technology in the Netherlands. Another related TFOT story is the usage of a common plant virus to deliver drugs specifically into cancer cells – allowing effective treatment without harming the surrounding tissue.
For more information about the drugs encapsulated in nanoparticles, see MIT’s official press release.