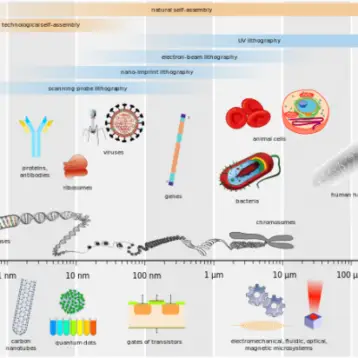
While the idea of using specific nano-particles to target tumors is not new, the nanoworms are a completely new invention, and have remarkable advantages over spherical nano-particles.
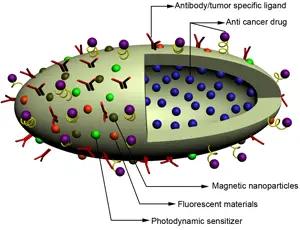
The researchers synthesized the nanoworms by coating spherical nano-particles of iron oxide with a polysaccharide called dextran. The coated nano-particles aggregated in a linear fashion, essentially creating ‘nanoworms’. Each nanoworm is 30 nano-meters long, making it about 3 million times smaller than an earthworm.
“The reason that these worms work so well is due to a combination of their shape and to a polymer coating on their surface that allows the nanoworms to evade these natural elimination processes,” said Michael Sailor, a Professor of chemistry and biochemistry at UC San Diego who led the research team. “As a result, our nanoworms can circulate in the body of a mouse for many hours.”
During their travels in the bloodstream, the nanoworms reach nearly every organ and tissue in the body. In order to make sure they concentrate mainly at the site of the tumor, the researchers coated the nano-worms with a tumor-specific targeting molecule. This peptide, called F3, which allows the nanoworms to home in on tumors, was developed in the laboratory of Erkki Ruoslahti, a cell biologist and professor at the Burnham Institute for Medical Research at UC Santa Barbara.
“Because of its elongated shape, the nano-worm can carry many F3 molecules that can simultaneously bind to the tumor surface,” said Sailor. “And this cooperative effect significantly improves the ability of the nanoworm to attach to a tumor.”
The scientists were able to verify that their nanoworms homed in on tumor sites by injecting them into the bloodstreams of mice with tumors. They found that the nanoworms had indeed aggregated in the tumors, although a major uptake of nanoworms by the liver was also noticeable. Unlike spherical nano-particles of similar size that were shuttled out of the blood stream by the immune system, the nanoworms remained mice’s in the bloodstream for hours.
“This is an important property because the longer these nano-worms can stay in the bloodstream, the more chances they have to hit their targets, the tumors,” said Ji-Ho Park, a UC San Diego graduate student in materials science and engineering, who is working in Sailor’s laboratory. Park was the motivating force behind the discovery when he found by accident that the gummy worm aggregates of nano-particles exhibited long-lasting retention time in the bloodstream.
Despite the good results, the researchers are well aware that the current design can still be improved. Since the nanoworms aggregate in the liver as well as at the tumor sites, they cannot be optimal carriers for the anti-cancerous drugs just yet. In order to deal with this problem, the researchers are currently working on other molecules and drugs that can be attached to the nanoworms and should increase their specificity and functionality.
“We are now using nanoworms to construct the next generation of smart tumor-targeting nano-devices,” said Ruoslahti. “We hope that these devices will improve the diagnostic imaging of cancer and allow pinpoint targeting of treatments into cancerous tumors.”
Other researchers involved in the development of the nanoworms are Michael Schwartz of UC San Diego, Geoffrey von Maltzahn of MIT, and Lianglin Zhang of UC Santa Barbara. The project was funded by grants from the National Cancer Institute of the National Institutes of Health.
TFOT has recently reported on drug-coated nano-particles, used to fight cancer more efficiently. Nano-particles were also surveyed as a catalyst for fuel-cell cars.
More information about the nano-worms can be found on the University of California, San Diego website.